Derived from the Greek words for sea (θάλασσα) and blood (αίμα), thalassemias are a group of inherited, genetic blood disorders. Thalassemias occur when the production of hemoglobin, a protein that carries oxygen within the red blood cells (RBCs) is disrupted. A life-threatening disease, thalassemia is an autosomal recessive condition with over 100,000 affected babies being born every year [1].
Hemoglobin consists of two parts called alpha and beta proteins. When the production of one of these proteins is affected, it results in either alpha (α) or beta (β) thalassemia. Severity depends on the type of thalassemia and whether one has the ‘minor’, ‘intermedia’, or ‘major’ form – defined by the number of genes that are altered. α-thalassemia is most commonly found in Africa, the Middle East, and India, while β-thalassemia is more prevalent in the Mediterranean.
Contents
Symptoms and treatment of thalassemia
Symptoms and treatment of thalassemia vary depending on the type and severity of the condition. In mild cases, symptoms might be non-existent and treatment is not required – carriers only have the risk of passing the affected gene to their offspring. In moderate to severe cases, symptoms may include fatigue, weakness, pale or yellowish skin, slow growth, abdominal swelling, dark urine, and facial deformities [2, 3]. Babies with the most severe cases may be born stillborn, die shortly after birth, or require lifetime treatment [2]. Untreated patients might show growth retardation, jaundice, poor musculature, skeletal changes, and severe anemia [2]. Lifelong transfusion therapies and iron chelation procedures to remove the excess iron that is accumulated are needed for moderate and severe cases. The excess iron in the body is one of the most severe complications of thalassemia treatment, as it can damage the heart, liver, and other internal organs [2, 4]. Additionally, spleen enlargement may occur due to the spleen overworking to remove all the damaged RBCs and avoid possible infections. While spleen removal is an option, it leaves the body more prone to infections. Other complications of thalassemia treatment include hypothyroidism, heart failure, liver and gall bladder problems, and diabetes [2, 4]. A bone marrow transplant to eliminate the need for lifelong blood transfusions might be a treatment option for severe cases, and gene therapy to correct the mutated gene could be applied in the future.
Genetic testing for thalassemia
Even though thalassemia is more common in specific countries, its prevalence is worldwide due to population migration and intermarriage between different ethnic groups. Thalassemia carriers do not exhibit any symptoms and may be unaware of their genetic status, but their children have a 50% chance of being carriers and a 25% chance of being affected. Therefore, thalassemia carrier screening is one of the most widely performed genetic tests, and countries that are most affected by thalassemia have developed their screening programs. These programs vary in being either mandatory or voluntary; being offered premaritally, preconceptionally, or prenatally; and on the public education and counseling provided. Examples of successfully implemented thalassemia screening programs are those of Italy and Cyprus. The region of Sardinia introduced a voluntary screening program in premarital and antenatal couples in 1975 which saw the thalassemia incidence fall from 1:250 to 1:4,000 in 20 years, a 95% incidence decrease [5]. A ‘quasi-mandatory’ premarital screening program in Cyprus, where 1 in 7 people is a carrier of β-thalassemia, was implemented in collaboration with the Greek Orthodox Church in the early 1980s [6]. The thalassemia laboratory tests couples free of charge and issues them a certificate to confirm the testing – which is required if the couples wish to be married by the church. Genetic counseling and information on reproductive options, like pre-implantation genetic diagnosis (PGD) or prenatal testing, is offered to them if they have a risk of having an affected child. This led to a significant decrease in the birth of affected individuals that had a beneficial role in improving the treatment of thalassemic patients – otherwise, there would have been a significant blood shortage on the island [7].
NIPT for thalassemia
In addition to carrier screening, prenatal detection of thalassemia is possible through non-invasive prenatal testing (NIPT), a simple and painless procedure that tests the fetus’s DNA through a maternal blood sample, and compares it to the maternal and paternal DNA to check for the genetic status. Nowadays, NIPT is routinely used for chromosomal aneuploidies as it provides the expecting parents with information early, reassures them, and allows them to make informed decisions. Its use in monogenic disorders, like thalassemia, is novel and provides an alternative option to the more expensive PGD. Confirmation of high-risk pregnancies by invasive procedures, like chorionic villi sampling or amniocentesis is recommended.
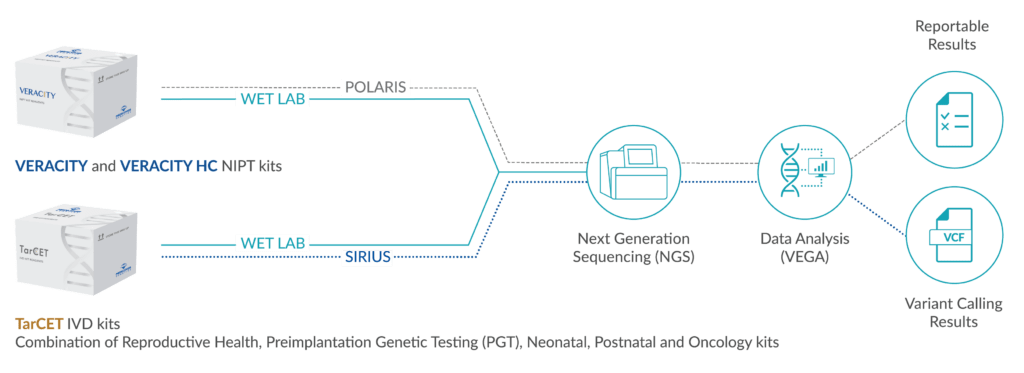
The VERAgene NIPT can detect thalassemia, along with other monogenic disorders, aneuploidies, and microdeletions from the 10th week of pregnancy. NIPT results, possible next steps, and clinical management should always be fully discussed with your healthcare provider.
References
[1] National Human Genome Research Institute. About Thalassemia. Retrieved 08 May 2019 from https://www.genome.gov/Genetic-Disorders/Thalassemia
[2] Mayo Clinic. Thalassemia. Retrieved 08 May 2019 from https://www.mayoclinic.org/diseases-conditions/thalassemia/symptoms-causes/syc-20354995
[3] Genetic and Rare Diseases Information Center. Thalassemia. Retrieved 08 May 2019 from https://rarediseases.info.nih.gov/diseases/7756/thalassemia
[4] Kids Health. Beta Thalassemia. Retrieved 08 May 2019 from https://kidshealth.org/en/parents/beta-thalassemia.html
[5] Mitchell JJ et al. Twenty-year outcome analysis of genetic screening programs for Tay-Sachs and beta-thalassemia disease carriers in high schools. Am J Hum Genet. 1996 Oct;59(4):793-8. PMID: 8808593; PMCID: PMC1914789. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1914789/
[6] Thalassaemia International Federation. Thalassaemia. Retrieved 08 May 2019 from https://thalassaemia.org.cy/about/thalassaemia/
[7] Cousens NE et al. Carrier screening for beta-thalassaemia: a review of international practice. Eur J Hum Genet. 2010 Oct;18(10):1077-83. doi: 10.1038/ejhg.2010.90. Epub 2010 Jun 23. PMID: 20571509; PMCID: PMC2987452.https://www.nature.com/articles/ejhg201090